BENTHIC FAUNA/COMMUNITIES
Published studies are reviewed/summarized and laid out below for five principle areas:
Figure 3, and the legend that accompanies it, indicate the locations of the reviewed studies.
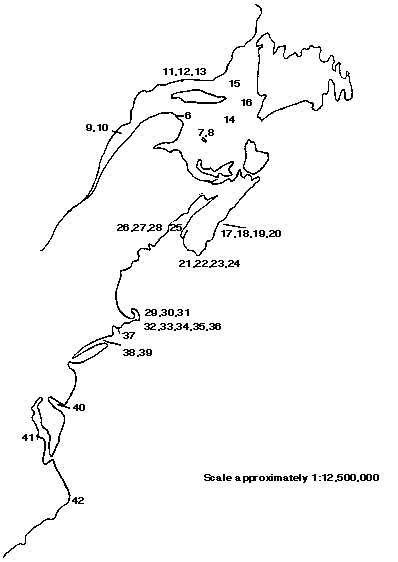
FIGURE 3: Locations of studies reviewed. See Legend on following page for locations and sources of studies.
Legend for Figure 3 providing literature sources and locations for marine benthic studies displayed in Figure 3.
|
STUDY
|
SOURCE
|
LOCATION
|
|
1 2 3 |
Caddy et al., 1977 Anonymous, 1997, 1998 Scarrat & Lowe, 1972 |
Northumberland Strait Northumberland Strait Kouchibouguac Bay, Northumberland Strait |
|
4 5 6 7 8 9 10 11 12 13 14 15 16 |
Hughes & Thomas, 1971a Hughes & Thomas, 1971b Brunel, 1971 Bourget & Messier, 1983 Hudon & Lamarche, 1989 Robert, 1979 Himmelman & Lavergne, 1985 Himmelman, 1991 Jalbert et al., 1989 Himmelman & Dutil, 1991 Peer, 1963 Long & Lewis, 1987 Wieczorek & Hooper, 1995 |
Bedeque Bay, Prince Edward Island Bideford River estuary, Prince Edward Island Bay of Gaspe, Gulf of St. Lawrence Magdalen Islands Magdalen Islands St. Lawrence Estuary St. Lawrence Estuary Mingan Islands Mingan Islands Mingan Islands Magdalen Shallows/Laurentian Channel Anticostic & Esqiman Channels Bonne Bay, Newfoundland |
|
17 18 19 20 21 22 23 24 |
Brawn et al., 1968 Miller et al., 1971 Peer, 1970 Volkaert, 1987 Elner & Campbell, 1987 Hatcher et al., 1998 Barbeau et al., 1996 Drummond-Davis et al., 1982 |
St. Margarets Bay, Nova Scotia St. Margarets Bay, Nova Scotia St. Margarets Bay, Nova Scotia St. Margarets Bay, Nova Scotia Southern Nova Scotia Southern Nova Scotia Southern Nova Scotia Southern Nova Scotia |
|
25 26 27 28 |
Caddy, 197 Wildish et al., 1972 Wildish, 1983 Logan et al., 1983 |
Bay of Fundy L'Etang Inlet, Bay of Fundy Passamoquoddy area, Bay of Fundy Passamoquoddy area, Bay of Fundy |
|
29 30 31 32 33 34 35 36 37 38 39 40 41 42 |
Dexter, 1944 Dexter, 1947 Young & Rhoads, 1971 Sanders, 1958 Sanders, 1960 Weiser, 1960 Wigley & McIntyre, 1964 Lee, 1944 Stickney & Stringer, 1957 MacKenzie, 1977 O'Connor, 1972 Maurer et al., 1978 Virnstein, 1977 Day et al., 1971 |
Ipswich Bay, Massachusetts Cape Ann, Massachusetts Cape Cod Bay, Massachusetts Buzzards Bay, Massachusetts Buzzards Bay, Massachusetts Buzzards Bay, Massachusetts Martha's Vineyard, Massachusetts Menemsha Bight, Massachusetts Greenwhich Bay, Rhode Island Long Island Sound, Conneticut Moriches Bay, Long Island, New York Delaware Bay, Delaware Chesapeake Bay, Maryland Cape Lookout, North Carolina |
3.1 NORTHUMBERLAND STRAIT
Northumberland Strait is that area between Prince Edward Island and the mainland of the provinces of New Brunswick and Nova Scotia, and has water depths of 10-20 m at the western end and >30 m at the eastern end (Kranck, 1972). The environmental conditions within the Strait are markedly different from those prevailing on the exposed Atlantic and lower Fundy shores (MacFarlane, 1966); conditions within St. Georges Bay however, are largely similar to conditions in the Strait (Moseley and MacFarlane 1969). Despite the intensive use of the area for fisheries by three provinces, and megaproject development (Confederation Bridge), little work has been done on the benthos in the Northumberland Strait area.
A survey of the benthic fauna of Northumberland Strait from the northwestern to eastern end of Prince Edward Island was conducted in 1975 with 96 stations being occupied taking samples from depths ranging from 5-49 m (the majority were 10-20 m deep) (Caddy et al., 1977). Bottom sediments were composed of gravel/coarser material, sand, and silt/clay. The Strait was divided into four areas (A-D) of which Area D, with a substrate of principally mud and mud-clay, is immediately adjacent to St. Georges Bay. Results are summarized here for both the entire Strait as well as Area D. Seventy four species of algae (43 species of Rhodophyta, 27 species Phaeophyta, 2 species each of Chlorophyta and Cyanophyta) were found with the most commonly occurring (>50% of samples) being Phyllophora spp., Polysiphonia spp., and Laminaria longicruris. Species found in 25-50% of samples included Ectocarpus siliculosus, Desmarestia aculeata, Fucus spp., Cystoclonium purpureum, Rhodomela confervoides, and Rhodymenia palmata.
Benthic macrofauna results from van Veen grab samples are presented in Table 1 including both the Strait as a whole and Area D. A complete species list of the 343 invertebrate species collected from this sampling program is presented in Appendix 3. Polychaetes indicate the greatest species abundance followed by amphipods, bivalves and arthropods, gastropods, and finally echinoderms. Sampling by beam trawl and scallop dredge indicated that echinoderms (Asterias vulgaris [64% of stations], Henricia sanguinolenta [53%], Echinarachinus parma [47%]) were the most widespread benthic fauna. Of 25 bivalve species sampled by trawl the mussels Modiolus modiolus (35.5% of stations) and Mytilus edulis (31.3%) were most common. Of 37 gastropod species the most commonly encountered were Nassarius trivittatus (21% of stations), Lunatia heros (17%) and Neptunea decemcostata (14%). The most common arthropods, excluding the prevalent amphipods, were Crangon septemspinosa (36% of stations), Pagurus acadianus (30%), and Pandalus montagui (21%). Other commonly encountered fauna included hydroids (51% of stations), bryozoans (47%) and Porifera (47%).
Table 1. Number of species, families, and most common species captured in Northumberland Strait in 1975 in van Veen grab samples. Area D is that portion of the Northumberland Strait study area immediately adjacent to St. Georges Bay. Data from Caddy et al. (1977).
|
Northumberland Strait
|
Area D
|
|||||
|
# of species
|
# of families
|
Most common species
|
# of species
|
# of families
|
Most common species
|
|
| Bivalves |
26
|
13
|
Asarte undata Clinocardium ciliatum Tellina agilis Thyasira gouldii |
19
|
11
|
Clinocardium ciliatum Modiolus modiolus Nucula tenuis Thyasira gouldii Yoldia sapotilla |
| Gastropods |
16
|
12
|
Nassarius trivittatus Retusa canaliculata |
5
|
4
|
Admete couthouyi Oenopta (Lora) elegans Oenopta turricula |
| Polychaetes |
91
|
29
|
Ninoe nigripes Pholoe minuta Prionospio steenstrupi |
58
|
25
|
Pholoe minuta Prionospio sttenstrupi Spio sp. |
| Arthropods |
26
|
13
|
Aeginina longicornis Diastylis quadraspinosa Diastylis sculpta Eudorella trunculata |
16
|
8
|
Diastylis quadraspinosa Eudorella emarginata Eudorella trunculata Leucon nasica |
| Amphipods |
73
|
19
|
Ampelisca macrocephala Ampelisca vadorum Corophium bonelli Phoxocephalus holbolli Unciola irrorata |
41
|
17
|
Ampelisca macrocephala Byblis gaimardi Corophium bonelli Harpinia propinqua Phoxocephalus holbolli |
| Echinoderms |
6
|
Echinarachinus parma Ophiura robusta |
5
|
5
|
Asterias vulgaris Ophiura robusta Stongylocentrotusdrobachiensis |
|
In terms of biomass, the echinoderms contributed the majority in the western half of Northumberland Strait (41.1-86.38% of total biomass). The polychaetes, bivalves and gastropods made up 13-28% of the biomass in this area and arthropods <2%. In Area D however, adjacent to St. Georges Bay, bivalves contributed greatest to the biomass (42.48%) followed by polychaetes (25.18%) echinoderms (15.13%), gastropods (2.32%), and arthropods (0.62%). Other species not falling into these taxa account for 14.26 %. Biomass of fauna (g wet weight/m2) for the whole Strait ranged from 0.01-750.6 for polychaetes, 0.01-112.6 for bivalves, 0.01-137.1 for gastropods, 0.01-555.2 for echinoderms, and 0.01-19.3 for arthropods. Within Area D total biomass (all taxa combined) ranged from 2.25-141.6 g wet weight/m2, with biomass of individual taxa ranging as: polychaetes (0.3-750.6 g wet weight/m2), bivalves (0.44-112.6), gastropods (0.21-123.2), echinoderms (0.01-418.1) and arthropods (1.52-608.1).
In 1967, Scarrat and Lowe (1972) conducted a study on the biology of the rock crab (Cancer irroratus) in Kouchibouguac Bay. Sampling between 4 and 18 m depth these authors found a standing stock ranging from 0.7 to 3.8 g/m2, with the greater values being found on boulders in both the spring and fall.
In 1980, Dunbar et al. (1980) compiled existing information to "describe and map the known distribution of the marine communities and faunal and floral elements in the Gulf of St. Lawrence". Benthic invertebrates and algae reported by these authors in the vicinity of the St. Georges Bay study area are listed in Table 2. Due to the paucity of information on the St. Georges Bay area, the mobility of invertebrates, and the prevailing currents from west through the Strait into St. Georges Bay, distribution information from Dunbar et al. (1980) was extracted from as distant as the eastern point of P.E.I. and westward to Pictou Harbour. Dunbar et al. (1980) emphasize that the blank areas on their charts from which this information is drawn are not reflective of absence of a given species but often these areas have not been sampled to determine presence. Thus, the list derived from their work is not to be construed as thorough or complete. Further sampling is required to establish presence and distributions of most components of the benthic flora and fauna.
Table 2. Species list for St. Georges Bay area and eastern Northumberland Strait from Dunbar et al. (1980).
|
Algae
|
Macroinvertebrates
|
||
| Phaeophycea |
Laminaria digitata |
Bivalvia | Arctica islandica Astarte subaequilatera Astarte undata Clinocardium ciliatum Crassostrea virginica Crenella glandula Macoma tenta Mercenaria mercenaria Mulina lateralis Mya arenia Mya truncata Nucula proxima Nucula tenuis Pandora glacialis Periploma leanum Petricola pholadiformis Pitar morrhuana Placopecten magellanicus Teredo navalis Yoldia limatula Volsella modiolus Yoldia sapotilla Yoldia thraciaeformis |
| Rhodophyta | Ceramium fastigatum Ceramium rubrum Euthora cristata Polysiphonia harveyi Polysiphonia urceolata Rhodymenia palmata Trailliella intricata |
Gastropoda | Acmaea testudinalis Admete couthouyi Buccinum undatum Crepidula fornicata Crepidula plana Nassarius trivittatus Polineces heros Polineces immaculata Urosalpinix cinerea |
| Chlorophyta | Enteromorpha linza Ulva lactuca |
Polyplacophora | Ischnochiton ruber Ischnochiton alba |
| Crustacea | Cancer irroratus Crangon septemspinosa Homarus americanus Pagurus acadianus |
||
Construction through the mid-1990s of the Confederation Bridge linking New Brunswick with Prince Edward Island resulted in the examination of benthic fauna and flora as part of an Environmental Impact Assessment. The two principle sampling methods were direct observation, by SCUBA and Remotely Operated Vehicles (ROV), and colonisation studies of organisms on the newly placed piers. The water depths ranged from 5 to 25 m and substrate was primarily cobble, shell, sand, and silt but with some bedrock and boulders. The following discussion is drawn from Anonymous (1997, 1998). Underwater video indicated only a few macrobenthic invertebrates. These were Homarus americanus, Cancer irroratus, Placopecten magellanicus, Pagurus sp., Metridium sp., Mytilus edulis, Asterias sp. Modiolus modiolus, hydrozoans and poriferans. Calculated densities based on this video for the prominent species were 0-0.04 individuals/m2 for H. americanus, 0-0.32/m2 for C. irroratus, 0-0.056/m2 for P. magellanicus, and 0-0.012/m2 for Pagurus sp. In a dredge disposal site in Amherst Cove, Homarus americanus densities were almost consistently zero animals/m2 between 1994 and 1997 though occasionally did rise to 0.6/m2. Cancer irroratus in this same time period ranged from 0 to 2.23 animals/m2, though 1/m2 may be more representative of maximum density.
The colonisation study of piers in Northumberland Strait (Table 3) indicate quite evenly distributed species numbers between the three phyla of algae and 11 phyla/classes of invertebrates. The bivalves, gastropods, polychaetes and crustaceans together only account for 54% of the 68 species recorded. The bryozoans, hydroids and poriferans also contribute significantly to the total species present (together accounting for 25% of species present). Unfortunately biomass was not recorded in this survey, instead presence was quantified by percent cover. On the piers the dominant faunal and floral groups in 1996-97 were (approximated from Figures 4.23-4.26 in Anonymous 1998):
0-5 m depth - Barnacles (20-60% cover), Enteromorpha (25-45% cover), algal mat (5-80% cover), bryozoans/hydrozoans (5-25% cover) and mussels (generally < 15% cover).
5-10 m depth - Barnacles (25-40% cover), algal mat (20-30% cover), bryozoans/hydrozoans (<5-45% cover), and sea anenomes (5 - 20% cover).
10-15 m depth - Barnacles (20-40% cover), algal mat (15-85% cover), bryozoans/hydrozoans (20-45% cover), sea anenomes (5 - 25% cover) and sea stars (0 - 25% cover).
15-20 m depth - Barnacles (10-45% cover), bryozoans/hydrozoans (10-55% cover) and sea anenomes (10 - 30% cover).
20-25 m depth - Barnacles (25-40% cover), bryozoans/hydrozoans (10-50% cover) and sea anenomes (5 - 35% cover).
>25 m depth - Barnacles (15 - 35% cover), bryozoans/hydrozoans (20-55% cover), algal mat (5-20% cover), and sea anenomes (5-30% cover)
Table 3. Species list of algae and invertebrates reported from two years (1996, 1997) sampling in Northumberland Strait in 5-20m of water as part of Confederation Bridge Project. Table compiled from Anonymous, 1997 9Talbes 5.7-5.100 and 1998 (Tables 4.2-4.12).
|
Algae
|
Invertebrates
|
||
| Phaeophyta | Asperococcus echinatus Chorda filum Chordaria flagelliformis Eudesme virescens Fucus serratus Saccorhiza dermatodea |
Bryozoa | Averillia sp. Bryozoa membranacea Bugula turrita Flustra foliacea Membranipora sp. Schizoporella unicornis |
| Chlorophyta | Chaetomorpha melangonium Chaetomorpha sp. Cladomorpha sp. Cladophora albida Cladophora seriacea Cladophora sp. Enteromorpha (2 species) |
Hydroid | Bougainvillia sp. Campanularia calceolifera Campanularia sp. Hydractinia sp. Sertularia argentea Sertularia sp. Tubularia crocea Tubularia sp. |
| Rhodophyta | Antithamnion sp. Ceramium (2 species) Ceramium rubrum Chondrus crispus Corallina offinalis Cystoclonium ceranoides Gelidium sp. Phyllophora pseudoceranoides Polysiphonia nigrescens Polysiphonia sp. Spermothamnion repens Spermothamnion sp. |
Nematode | Nematoda |
| Porifera | Halichondria bowerbanki Scypha ciliata Scypha sp. |
||
| Anthozoa |
Metridium senile |
||
| Bivalvia | Anomia simplex Gammarus sp. Hiatella arctica Modiolus modiolus Modiolus sp. Mytilus edulis Mytilus sp. Spisula solidissima |
||
| Gastropoda | Aeolidia papillosa Coryphella sp. Crepidula convexa Crepidula fornicata Crepidula plana Dendronotus frondosus Eubranchus sp. Facelina bostoniensis Lacuna vincta Littorina sp. Mitrella lunata Nassarius trivittatus Notoacmaea testudinalis Onchidoris |
||
| Polychaeta | Eulalia viridis Eusyllis blomstrandi Gattyana cirrosa Harmothoe sp. Neries sp. Polydora ciliata Phyllodoce sp. |
||
| Crustacea | Aeginella longicornis Balanus balanoides Balanus sp. Cancer irroratus Caprella linearis Caprella sp. Corophium volutator Homarus americanus Jassa falcata Pagurus acadianus Pagurus pubescens |
||
| Pycnogoda | Phoxocilidium femoratum | ||
| Echinodermata | Asterias forbesi Asterias vulgaris Henricia sp. |
||
| Tunicata | Thaiacea (salp) Doliolum sp. |
||
Summary of Northumberland Strait
Based on the reported sampling from Northumberland Strait between 1967 and 1998 it appears that polychaetes are present in the greatest species abundance (up to 91 species) followed by amphipods (up to 73 sp.), bivalves (up to 26 sp.), gastropods (up to 16 sp.), and non-amphipod crustaceans and echinoderms each up to 6 species present. Algal species are present in the range of 3-27 species (Phaeophyta), 7-43 species (Rhodophyta), and 2-8 species (Chlorophyta).
Biomass ranges up to 1,400 g/m2 with bivalves, gastropods, polychaetes and echinoderms making up the majority of the contribution. The biomass is generally greatest for the larger organisms (echinoderms and bivalves), with lower contributions by the polychaetes, gastropods and arthropods. However, this large contribution by molluscs and echinoderms includes inedible tests/skeletons etc., and so it is questionable how relevant these biomass estimates are from a trophic level perspective, as much of the greatest biomass contribution may not be passed on to consumers. Common invertebrate species reported in Northumberland Strait (from Tables 1-3) include 31 bivalves, 23 gastropods, 11 polychaete species, 25 arthropods and 6 echinoderms. The most commonly encountered species in this limited sampling of Northumberland Strait were:
Bivalves: Modiolus modiolus, Mytilus edulis, Placopecten magellanicus
Gastropods: Nassarius trivittatus, Lunatia heros, Neptunea decemcostata
Arthropods: Crangon septemspinosa, Pagurus acadianus, Pandalus montagui, Homarus americanus, Cancer irroratus
Echinoderms: Asterias vulgaris, Henricia sanguinolenta, Echinarachinus parma
3.2 GULF OF ST. LAWRENCE
The fauna and subtidal communities of the Gulf of St. Lawrence are poorly known (Robert, 1979; Long and Lewis, 1987), except perhaps in qualitative or semi-quantitative terms (Bourget and Messier, 1983). The Gulf is a very large embayment composed of many different localized environments with differing factors (e.g., depth, temperature, salinity, substrate, etc.) affecting benthic community organization and structure. There appears to be different community structures and controls between the southern and northern Gulf; in the southern Gulf decapod crustaceans and fishes are thought to be influential in controlling community structure, while their absence in the northern Gulf results in whelks and seastars filling these predatory roles (Himmelman, 1991). The fauna and flora of the estuary itself are subarctic and boreal, more closely resembling the north shore of the Gulf and the Labrador coast than the southern Gulf and Maritimes (Himmelman and Lavergne, 1985). These differences must be kept in mind in the descriptions of the various studies below.
3.2.1 Southern Gulf
Sampling was conducted in the Bay of Gaspe between 1956 and 1960 involving 91 stations at depths of 9-100 m on substrates of sand, muddy sand, and mud (Brunel, 1971). Nineteen polychaete species were reported (two most common: Harmothoe extenuata and Gattyana cirrosa), as well as 11 bivalve species (most common: Serripes groenlandica, Clinocardium ciliatum, Spisula polynyma), 17 decapod crustaceans (most common: Sabinea septemcarinata, Argis dentata, Eualus macilentus), and 12 echinoderms (Ophiuri sarsi and Asterias vulgaris most common). The author recognized eight communities within this area; these are presented with their species in Table 4.
Table 4. Communities recognized in the Bay of Gaspe, 1956-1960. Data from Brunel (1971).
|
Euryboreal Soft Mud
|
Euryboreal Sand
|
Euryboreal Mixed Ground
|
|
Casco bigelowi
Chiridotea tuftsi Diastylis polita Nephthys incisa Pholoe minuta Phyllodoce mucosa |
Asteria vulgaris
Bostrichobranchus pilularis Chiridotea tuftsi Crangon septemspinosa Diastylis polita Echinarachinus parma Edotea triloba Hippomedon serratus Photis macrocoxa Phoxocephalus holbolli Phyllodoce mucosa Spisula polynyma Tmetonyx nobilis |
Ophiopholis aculeata
Ophiura robusta Strongylocentrotus drobachiensis |
|
Subarctic Muddy Sand
|
Subarctic Sandy Mud
|
|
|
Cerastoderma pinnulatum
Clinocardium ciliatum Leptocheirus pinguis Macoma calcarea Ophiura sarsi Serripes groenlandica Sternaspis scutata Thyasira gouldii |
Aporrhais occidentalis
Gattyana cirrosa Harmothoe extenuata Harmothoe imbricata Harmothoe nodosa Macoma calcarea Maldane sarsi Mya truncata Nuculana pernula Ophiura sarsi Priapulus caudatus Sabina septemcarintat Serripes groenlandica Steguphiura nodosa Yoldia norvegica |
|
|
Arctic Cohesive Mud
|
Arctic Sandy & Pebbly Mud
|
Arctic Mixed Ground
|
|
Arrhis phyllonyx |
Boltenia ovifera
Ophiura sarsi |
Chlamys islandica
Ophiocantha bidentata Ophiopholis aculatea Ophiura robusta Ophiura sarsi Strongylocentrotus pallidus |
In a classification and ordination exercise, Hughes and Thomas (1971a) delineated six groups within a Bedeque Bay, Prince Edward Island estuary from samples collected in 1967 from depths of 0.3 to 5.4 m (substrate description not provided). However, for the purposes of this paper only their first division, into euryhaline and lower estuarine species, is relevant. Their further divisions are of fine scale and statistical divisions; their biological or field assessment would be problematic in trying to define other areas with respect to these communities due to great variability in environmental and biological conditions. Their species groupings are presented in Table 5. These authors reported 62 species of which 20 are polychaetes, 14 bivalves, 9 gastropods, 6 crustaceans, 1 echinoderm and 8 algae species accounting for 93.5% of the total species number. Dominant species (i.e., > 50 individuals/m2 or >50 g dry weight/m2 for plants) at this location were Mytilus edulis, Tellina agilis, Nassarius obsoletus, Nassarius trivittatus, Littorina littorea, Crepidula plana, Lunatia heros, Polycirrus eximius, Neopanpoe texana, Balanus improvisus, Nemertea, Ulva lactuca, Chondrus crispus, Gracilaria verrucosa, and Zostera marina.
Table 5. Large scale division of estuarine species from Bedeque Bay, Prince Edward Island, 1967. Table modified from Hughes and Thomas (1971a).
|
Euryhaline
|
Lower Estuarine
|
||
| Bivalvia | Crassostrea virginica Mulinia lateralis Mytilus edulis Petricola pholadiformis Venus mercenaria |
Bivalvia | Ensis directus Mya arenia Pitar morrhuana Tellina agilis |
| Gastropoda | Crepidula fornicata Crepidula plana Littorina littorea Nassarius obsoletus |
Gastropoda | Lunatia heros Nassarius trivittatus |
| Polychaeta | Glycera americanus Harmothoe imbricata Nereis virens Notomastus latericeus Scoloplos fragilis |
Polychaeta | Glycera dibranchiata Maldanopsis elongata Nephthys incisa Pectinaria gouldii Pectinaria granulata Pherusa affinis |
| Crustacea | Balanus improvisus Cancer borealis Crangon septemspinosa Neopanope texana |
Crustacea | Pandora gouldiana |
| Plantae | Chondrus crispus Enteromorpha prolifera Gracilaria
verrucos Phyllophora sp. Polysiphonia grescens Ulva lactuca Zostera marina |
Amphipoda | Leptocheiros pinguis |
| Echinodermata | Asterias vulgaris | ||
In an analysis of a second Prince Edward Island estuary (Bideford River; sampling date not provided), Hughes and Thomas (1971b) collected benthic samples along four transects ranging in depths from intertidal to 4.7 m and substrates ranging from silt-clay to coarse sand (median particle size range 0.0085-0.79 mm). The most commonly captured invertebrates are provided in Table 6.
Table 6. Ranges of densities and biomass of common invertebrates (>20/m2 for transect 1-3; >100/m2 for transect 4) from Bideford River estuary, Prince Edward Island. Data from Hughes and Thomas (1971b).
|
Species
|
Density (/m2)
|
Biomass (g dry weight/m2) | |
| Bivalvia | Cumingia tellinoides Gemma gemma Macoma balthica Modiolus demissus Mya arenia Mytilus edulis Tellina agilis |
2-300
5-29,120 1-286 1-42 1-565 1-273 1-438 |
0.01-2.33
0.01-16.32 0.01-6.47 0.37-18.02 0.01-189.97 0.47-157.28 0.01-1.03 |
| Gastropoda | Crepidula fornicata Crepidula plana Littorina saxitalis Nassarius obsoletus Odostomia sp. Retusa canaliculata |
1-688
1-334 1-1,500 1-1,557 15-441 10-300 |
0.01-25.4
0.01-7.09 0.02-6.0 0.02-37.76 0.01-4.97 0.01-0.68 |
| Polychaeta | Clymenella torquata Harmothoe imbricata Nereis succina Nereis virens Notomastus latericeus Pectinaria gouldii Scalopolos fragilis |
2-780
2-260 2-134 1-790 12-875 2-264 1-170 |
0.01-3.59
0.01-1.04 0.04-0.49 0.01-11.33 0.01-0.52 0.02-0.59 0.01-0.59 |
| Amphipoda | Corophium insidiosum Gammarus locusta |
30-570
1-120 |
0.01-0.22
0.01-0.19 |
| Isopoda | Leptochelia rapex |
100-375
|
0.01-0.06
|
| Unclassified | Mysella planulata |
40-375
|
0.02-0.18
|
A Magdalen Island lagoon (maximum depth 8 m) was sampled in 1975 for macrobenthos density and biomass (Bourget and Messier, 1983). The substrate was predominantly sands with minor amounts of silt, clay and gravel (median particle diameter 0.25 mm). Fifty four benthic species were reported with a mean subtidal density of 3,398.1 individuals/m2 and mean biomass of 6.4 g/m2. The most common (i.e., present in highest density) organisms were the bivalve Gemma gemma (1,398.7 /m2), the gastropods Littorina saxatillis (1,345.6 /m2), Cingula aculeus and Hydrobia minuta (526.7 and 524.8 /m2 respectively), and the crustacean Corophium insidiosum (771.2 /m2). All other species were present at less than 500 individuals/m2. Table 7 presents ranges of values for individual densities and biomass for this area.
Table 7. Ranges of individual species densities and biomass grouped by Phyla/class for Magdalen Islands sampled in 1975. Data from Bourget and Messier (1983).
|
Individual Density (/m2)
|
Biomass (g dry wt./m2)*
|
|
| Sipuncula |
1.6-1.8
|
|
|
Mollusca
|
1.0-1345.6 |
0.578-1.249 (13.1-14.4) |
| Polychaeta |
1.1-92.5
|
2.2-3.943 (33.6-62.1)
|
| Crustacea |
1.0-771.2
|
0,1-2,151 (2.3-24.9)
|
* range in brackets is percent biomass
The Magdalen Island lagoons were sampled again in 1985 (Hudon and Lamarche, 1989), this time by SCUBA. Lobster (Homarus americanus) densities were found to range from 0.018-0.975 lobster/m2 and rock crab (Cancer irroratus) from 0.008 to 3.358 crab/m2. Four major substrate types were identified and the associated flora and fauna are listed below:
Small stones imbedded in soft sediments - Algae - Ptilota serrata, Chordaria tomentosa, Corallina officinalis. Invertebrates - Mytilus edulis, Modiolus modiolus, polychaetes (particularly Lepidonotus squamatus), littorinid gastropods, hydroids, barnacles, brittle stars.
Bedrock (sandstone) - similar to above
Piles of large rocks - Algae -Laminaria spp., Ulva spp., Chondrus crispus, Chordaria tomentosa, Rhodomenia palmata. Invertebrates - Caprellid amphipods, Strongylocentrotus drobachiensis, gastropods (Lacuna vincta, Nassarius sp.), Mytilus edulis, Modiolus modiolus, hydroids, ascidians, Porifera, nereid polychaetes.
Sand - Zostera marina, Ulothryx. Invertebrates - Echinarachinus parma, polychaetes, burrowing bivalves (Venus mercenaria, Ensis directus, Mya arenia, Crangon septemspinosa).
3.2.2 Northern Gulf
In 1962 benthic sampling was undertaken in the northern Gulf of St. Lawrence along the northeast edge of the Magdalen Shallows and the Laurentian Channel (Peer, 1963). Two of the stations (Stations 26 & 78) were within the depth range of the St. Georges Bay study area; at depths of 73 and 86 m respectively. Station 26 had a substrate of unsorted gravel and Station 78 substrate was fine sand. Thirty one taxa were identified from Station 26, with Yoldia myalis (15.5%), Strongylocentotus droebachiensis (13.9%), polychaetes (12.1%), and Gephryea (11.7%) each contributing the greatest presence. Pagurus spp. (9.0%), Ophiuridea (7.3%), and Coelenterata (7.1%) contributed between 5 and 10% each, and the remaining 22 taxa each contributed <5% to the standing crop. Estimated biomass at this station was 6.16 g dry organic matter/m2. Seventeen taxa at this site made up 95% of the biomass. Station 78, on fine sand, was dominated by the sand dollar Echinarachinus parma (78.8% of standing crop) with much lesser contributions by Ophiuroidea (9.3%) and 28 other taxa. Estimated biomass at Station 78 was 21.59 g organic matter/m2. At this station 95% of the biomass was represented by five taxa (E. parma, Ophiuroidea, Priscillina armata, Pectinaria spp., and Macoma balthica).
Long and Lewis (1987) report results of a 1981 sampling program in Anticosti and Esquiman Channels in the northern Gulf. Sample depths ranged from 37 to 285 m; only results from less than 150 m depth are included in Table 8. The substrate at the stations sampled was largely sand (0.062-1.0 mm) and gravel (1.0-64 mm), with some (generally <35%) pelite (<0.062 mm). They report 73 taxa collected when all of their sites (including the deep stations) are included of which 25 are molluscs, 22 are polychaetes, 9 amphipods, 4 echinoderms and the remainder as Cumacea (3 taxa), Cnidaria (3 taxa) and Other Phyla (6 taxa). They found that, in general, benthic diversity was maximum between 75 and 94 m and appeared to be highest on shelf/slope breaks and decreased to moderate levels on slopes. This is in agreement with Robert (1979) who found in a cluster analysis of environmental and biological variables in the St. Lawrence Estuary that there is a complete distinction between their sampled stations above and below 75 m depth. Robert (1979) also reports the shallower stations having the greatest diversity. However, though diversity decreased on slopes, abundance was high (Long and Lewis, 1987). Within depths representative of the St. Georges Bay study area the range of substrate component values in the study of Long and Lewis (1987) was from 5-98% pelites, 8-71% sand, and 4-32% gravel. Macrobenthos abundance ranged from 640 to 5,250 individuals/m2 and diversity (Shannon Weaver H') from 0.746-1.234.
Table 8. Depth, abundance and diversity/evenness indices of samples from the Anticosti and Esquiman Channels, 1981. Table modified from Long and Lewis (1987).
|
Depth (m)
|
Abundance (/m2)
|
Diversity Index (H')*
|
Evenness Index (J)
|
|
37 |
1,030
2,875 640 3,750 1,685 2,160 1,365 750 730 2,310 3,056 2,540 1,596 1,896 5,250 2,932 3,775 4,120 672 |
1.089
1.092 1.212 1.299 1.205 1.132 1.029 1.234 0.746 1.114 1.132 0.071 1.205 1.078 1.226 1.176 0.993 1.129 0.984 |
0.236
0.193 0.292 0.219 0.278 0.211 0.299 0.286 0.174 0.208 0.211 0.239 0.252 0.247 0.196 0.243 0.171 0.188 0.269 |
Himmelman (1991) provides qualitative description of the communities to depths of 20 m on four different substrate types from the Mingan Islands, northern Gulf of St. Lawrence. Macrobenthic species and principle algal species reported are presented below for three of his descriptions - 1) Moderately exposed, medium sloped bottoms, 2) Rocky faces; and 3) Gently sloping sediment bottoms in areas of strong tidal currents. These areas are thought to physically represent some of the areas within the St. Georges Bay study area though the previous caveat about differences in biotic communities between the southern and northern Gulf must be borne in mind.
Moderately exposed, medium sloped bottoms
- 4-6 m depth to bottom of rocky zone - Ophiopholis aculeata, Halocynthia pyriformis, Metridium senile, Tealia felina, Psolus fabricii and Cucumaria frondosa. Algal species Alaria sp., Saccorhiza dermatodea, Laminaria digitata, L. longicruris, Agarum cribrosum
- sediment bottoms below rocky slopes - Echinarachinus parma, Clinocardium ciliatum, Serripes groenlandica, and Spisula polynyma.
Rocky faces
- Ophiopholis aculeata, Halocynthia pyriformis, Metridium senile, Tealia felina, Myriopora subgrucila, Pallina sitiens.
Gently sloping sediment bottoms in areas of strong tidal currents
- Gersemia sp., Cucumaria frondosa, Echinarachinus parma, Psolus fabricii, Boltenia ovifera, Halocynthia pyriformis, Metridium senile, Lurcenaria quadricornis, Chlamys islandica, Tealia felina, Gorgonocephalus arcticus, Ophiura sarsi, Buccinum undatum, Aporrhais occidentalis. Algal species Agarum sp., Ptilota sp.
Himmelman (1991) also reports densities for predatory invertebrates in the Mingan Island area. The whelk Buccinum undatum had the highest density (0.417 /m2), four seastar species (Leptasterias polaris, Asterias vulgaris, Crossater paposus, and Solaster endeca) ranged from 0.002 to 0.15 /m2 and the crabs Hyas araneus (0.055 /m2) and Cancer irroratus (0.053 /m2) were also present at low densities. The gastropod Neptunea despecta tornata was only present at a density of 0.006 /m2. During 1982-1983, in this area the mean density of Buccinum undatum was found to range from 1.25 to 2.86 individuals/m2 between 0 and 20 m water depth with biomass estimates including 1.2 g/m2 (juveniles), 6.2 g/m2 (immatures) and 23.0 g/m2 (mature animals) (Jalbert et al., 1989). Densities of other species reported by Jalbert et al. (1989) are provided in Table 9. The percentage occurrence of various species by substrate are provided in Table 10. In this area the urchin Strongylocentratus drobachiensis is the most abundant organism on all substrates, with four each of bivalve and gastropod species, two other echinoderms, and a single polychaete species forming the remainder of the most commonly occuring species. Based on 1984 SCUBA sampling at the Mingan Islands, Himmelman and Dutil (1991) report the seastars Asterias vulgaris and Leptastarias polaris at densities <0.25/m2 at depths below 2 m (though up to 1.71/m2 in depths of 0-2 m). Crossater papposus is consistently below 0.03 individuals/m2. These results must be qualified, however, as Himmelman and Dutil (1991) point out that the reduced number of fish in the northern Gulf acting as predators relative to the Maritimes, and the resultant importance of seastars and whelks, suggest major differences in community structure between the two areas.
Table 9. Estimated densities of selected invertebrates from Mingan Islands, Quebec, 1982-83. Data from Jalbert et al. (1989).
| Species |
Depth (m)
|
Density (/m2)
|
|
Gastropoda |
14-20 <10 |
0.22-0.27 0.01-0.06 |
|
Crustacea Pagurus sp. |
0-8 8-20 0-2 18-20 |
0.06-0.08 <0.045 1.13 4.94 |
|
Echinodermata Crossater paposus Solaster endeca |
4-8 12-20 0-20 4-8 16-20 0-20 |
0.19 0.01-0.03 0.01 0.74 0.06 0.002 |
Table 10. Occurrence (percentage presence in sampled quadrats) of various invertebrate and algal species on various substrates near the Mingan Islands, 1982-83. Table from Jalbert et al. (1989).
| Species |
Bedrock
|
Boulders
|
Cobbles
|
Gravel
|
Sand-Mud
|
| Bivalvia Chlamys islandica Hiatella arctica Mya truncata Mytilus edulis Other large clams |
0 47.3 11.3 19.4 0.4 |
1.8 22.9 17.3 21.8 2.1 |
10.9 8.3 39.8 10.2 8.3 |
1.0 6.9 32.7 6.9 7.9 |
0.9 1.7 47.2 2.7 24.3 |
| Gastropoda Acmeae testudinalis Littorina sp. Margarites sp. Tonicella sp. |
40.3 14.5 17.3 38.9 |
73.6 11.6 34.5 46.1 |
53.0 12.4 18.8 38.7 |
45.5 5.0 4.0 31.7 |
17.7 2.7 6.1 9.4 |
| Polychaeta Pectinarea granulata |
6.7 |
9.9 |
28.2 |
10.9 |
13.2 |
| Echinodermata Echinarachinus parma Ophiopholis aculeata Strongylocentrotus drobachiensis |
0 75.3 95.8 |
2.8 57.0 92.6 |
11.7 43.2 94.7 |
21.8 9.9 87.1 |
29.9 9.7 87.2 |
| Algae Agarum cribrosum Alaria esulenta Ptilota serrata |
20.8 7.4 34.3 |
14.8 6.0 7.0 |
9.4 3.0 16.9 |
5.9 2.0 5.0 |
1.7 0.4 9.8 |
Sampling in the St. Lawrence estuary from 1970-72 is reported for mollusc species by Robert (1979). Sampling at stations between 15 and >365 m depth over sediments of silt-sand, silt-clay, and sand-silt (median particle diameter <0.063 - 0.25 mm) resulted in 52 mollusc species (Bivalvia = 32 species, Gastropoda = 16, Scaphapoda = 2, Polyplacophora = 1, Aplacophora = 1 species). Combined, the bivalves and gastropods represented 92% of the total mollusc species present. As stated previously, Robert (1979) found the highest diversity in shallow water and also suggests that sediment type is a more precise determinant than water temperature with respect to organism presence/absence. Robert (1979) also found that when diversity is high, the dominant molluscan species is not exceedingly abundant (<200 individuals) and is always a suspension feeder. He clustered mollusc 'assemblages' by species present and environmental variables (temperature, substrate) and two of the resultant clusters may be relevant to the St. Georges Bay study area (Table 11) despite the differences between the northern Gulf and the Maritimes. The number of species and individuals per sample, and diversity indices, are generally lower for the deeper group than the shallow water group.
Table 11. Range of diversity and eveness indices (Group A, n=13, Group F, n=10) of molluscan fauna in two depth ranges from the St. Lawrence Estuary, 1970-1972. Table adapted from Robert (1979).
|
Group A (<75m depth)
|
Group F (86-165m depth)
|
|
| Number of species per sample (S) |
15-34
|
8-18
|
| Number of individuals per sample (N) |
121-1140
|
64-631
|
| Brillions diversity index (H) |
0.18-0.98
|
0.1-0.59
|
| Shannon Weaver diversity index (H') |
0.17-0.99
|
0.1-0.66
|
| Hmax |
1.17-1.53
|
0.9-1.26
|
| Evenness (J) |
0.14-0.74
|
0.11-0.57
|
Hmax=log(S)
Himmelman and Lavergne (1985) identify four subtidal zones in the St. Lawrence Estuary based on 111 SCUBA diving transects. Their results are on rocky, shallow (<12 m) shorelines and their zonation is presented in Table 12. The Fringe Algal Zone extends from the low intertidal to a depth up to 4 m below Lowest Water at Spring tides (LWST). The Zone of Grazing-Resistant Algae is immediately below this, and at greater depth is the Barren Zone; an area lacking in species and unproductive. Finally, the Zone of Filter Feeders is found in the St. Lawrence Estuary at depths where the urchin density is much reduced, generally 4-10 m depth. The depths of these zones, if present in the St. Georges Bay study area, are unlikely to coincide with the depths reported here as the local conditions are different between the St. Lawrence estuary and the west coast of Nova Scotia.
Table 12. Characteristic species of the subtidal zonation of the St. Lawrence Estuary shallow water (<12 m) zones. Table modified from Himmelman and Lavergne (1985)
| Algae | Invertebrates | |
| Fringe Algal Zone | Alaria esculanta Chordaria flagelliformis Desmarestia aculeata Halosaccion ramentaceum Laminaria spp. Palmaria palmata Petalonia fascia Polysiphonia urceolata Porphyra spp. Rhodomela confervoides Saccorhiza dermatodea Spongomorpha arcta Ulvaria obscura |
Acmeae testudinalis Caprella spp. Gammarid amphipods Lacuna vincta Margarites costalis Margarites groenlandica Mytilus edulis Tonicella spp. |
| Zone of Grazing-Resistant Algae | Agarum cribrosum Callophylis cristata Clathromorphum spp. Desmarestia viridis Lithothamnion spp. Phycodrys rubens Ptilota serrata |
Acmeae testudinalis Strongylocentrotus droebchiensis Tonicella spp. |
| Barren Zone | Clathromorphum spp. Desmarestia viridis Lithothamnion spp. |
Acmeae testudinalis Hiatella arctica Strongylocentrotus droebchiensis Tonicella spp. |
| Zone of Filter Feeders | Clathromorphum spp. Lithothamnion spp. Peyssonnelia rosenvingii |
Ascidia spp. Boltenia ovifera Chondractinia tuberculata Cucumaria frondosa Didendium candidum Ectoprocts Halocynthia pyriformis Hydroids Metridium senile Ophiopholis aculeata Psolus fabricii Porifera Strongylocentrotus droebachiensis Tealia feline |
In Bonne Bay, Newfoundland, sampling in 1990 of substrate at depths of 44 to 212 m indicated that annelids were the most abundant group in terms of both abundance and biomass (Wieczorek and Hooper, 1995). The substrate at these sampling locations was composed of fine sand, very fine sand, and silt/clay. Two of these stations (South Arm and Wigwam Point) were less than 150 m deep and so the following is a discussion of these stations only as they are most relevant to the depths found in St. Georges Bay (Table 13). The annelids made up the greatest contribution to the faunal density, comprising 43-66% of the total invertebrates (the sedentary polychaetes formed 28.7-50% of total density), with crustaceans contributing 21.7-30% and the molluscs from 9.8-21%. The remaining groups each contributed less than 2% to the faunal density. Biogenic material (living and dead plant material, mollusc shells, polychaete mucous tubes and casings, fish bones and scales, and dead animal remains) made up by far the largest mass of organic matter within the substrate (56.7-84.5% of total). In general, the annelids contributed the greatest 'living' mass (12-20% of total) with the cnidarians providing a large contribution at Wigwam Point (17%). Contributions by the other groups to wet mass was variable between stations.
Table 13. Density and wet mass from sampling at two stations in Bonne Bay, Newfoundland, 1990. Note. Data has been converted to /m2, it is originally reported as /13,000 g substrate (sampled with a 0.1 m2 Petersen grab). Table adapted from Wieczorek and Hooper (1995).
|
South Arm
|
Wigwam Point
|
|||
| Depth (m) Dominant particle Diameter |
44-150
<0.062-0.25 |
68-126
<0.062-0.125 |
||
|
Density (/m2)
|
Wet Mass (g/m2)
|
Density (/m2)
|
Wet Mass (g/m2)
|
|
| Mollusca Bivalvia Cerastoderma spp. Small, thin shelled bivalvia Gastropoda Small gastropoda Scaphopoda |
81
9 0 6 53 53 19 |
45
11 0 1 32 32 2 |
129
67 3 52 41 40 22 |
51
35 12 5 13 12 3 |
| Annelida Sedentary, burrowing and tubiculous Polychaeta Errant surface dwelling Polychaeta Errant burrowing Polychaeta |
542
411 53 2.8 |
732
551 109 5.5 |
267
176 24 26 |
343
238 42 45 |
| Crustacea Amphipoda Small crustacea |
178
156 22 |
49
46 3 |
187
163 24 |
45
43 2 |
| Echinodermata Ophiuroidea Asteroidea Holothuroidea |
3
0 1 2 |
82
0 80 2 |
2
1 0 1 |
4
1 0 3 |
| Cnidaria Colonial Hydrozoa Anthozoa Porifera |
8
6 0 1 |
6
3 0 1 |
12
10 2 4 |
293
1 292 1 |
| Other invertebrates |
9
|
31
|
4
|
8
|
| Total invertebrates (/m2) |
820
|
|
613
|
|
| Total mass (g/m2) |
|
6,105
|
|
1,725
|
Summary of Gulf of St. Lawrence
This review of the benthos of the Gulf of St. Lawrence clearly indicates the difference between the southern and northern Gulf. The results are summarized below.
Based on the limited sampling reported for the southern Gulf (five studies) the bivalves, gastropods and polychaetes appear to be the dominant organisms with up to 14 bivalve species, 9 gastropod species and 20 polychaete species being reported within single studies. In terms of biomass, bivalves appear to contribute to a greater degree than gastropods, Polychaetes are variable, ranging from the greatest contributor of biomass (Bourget and Messier, 1983) to the least of the three groups (Hughes and Thomas 1971b).
Seven reported studies from the northern Gulf of St. Lawrence and the estuary produce different results from the southern Gulf. Crustaceans are a more significant component of the presence, being in greater abundance than molluscs and echinoderms in one study. Individual species appear to dominate the benthos to a greater degree in the northern Gulf (e.g., Buccinum undatum, Strongylocentrotus drobachiensis, Yoldia myalis, Asteria sp.). Polychaetes, however, continue to contribute significantly to the benthic community in the northern Gulf of St. Lawrence.
Perhaps the greatest contrast between the southern and northern Gulf is in the 'Dominant' species collected (i.e., most commonly encountered or abundant per capture event). See Table 14 for species comparison. As may be readily seen, the benthic composition of the two areas are significantly different. The number of common species per phyla/class for the most common groups range from 8-10 for bivalves, 7-11 for gastropods, 1-11 for polychaetes, and 2-10 for echinoderms. Decapod crustaceans are conspicuously absent from this list.
Table 14. Listing of 'dominant' species reported from the southern and northern Gulf of St. Lawrence. Absence of species from the table does not imply that it is absent, only that it is present at lower density/abundance than more dominant species. (e.g., Buccinum is present in southern Gulf, but not commonly reported as abundant or dominant species). Table compiled from studies reviewed herein.
|
Southern Gulf
|
Northern Gulf
|
|
| Bivalvia | Clinocardium ciliatum Cumingia tellunoides Gemma gemma Macoma balthica Modiolus demissus Mya arenia Mytilus edulis Serripes groenlandica Spisula polynyma Tellina agilis |
Chlamys islandica Clinocardium ciliatum Hiatella arctica Mya truncata Mytilus edulis Serripes groenlandica Spisula polynyma Yoldia myalis |
| Gastropoda | Cingula aculeus Crepidula fornicata Crepidula plana Hydrobia minuta Littorina littorea Littorina saxitalis Lunatia heros Nassarius obsoletus Nassarius trivittatus Odostomia sp. Retusa canaliculata |
Acmae testudinalis Aporrhais occidentalis Buccinum undatum Lacuna vincta Littorina sp. Margarites sp. Neptunea despecta |
| Polychaeta | Clymenella torquata Gattyana cirrosa Harmothoe extenuata Harmothoe imbricata Lepidonotus squamatus Nereis succina Nereis virens Notomastus laticerus Pectinaria gouldii Polycirrus eximius Scalopolos fragilis |
Pectinarea granulata |
| Echinodermata | Asterias vulgaris Ophiuri sarsi |
Asteria vulgaris Crossaster papposus Cucumaria frondosa Echinarachinus parma Gorgonocephala arctica Leptastarias polaris Ophiopholis aculatea Psolus fabricii Solaster endeca Strongylocentrotus drobachiensis |
| Total Species |
34
|
26
|
| Number of Species in Common |
6
|
|